Effects of an Evidence-based Exercise Intervention on Clinical Outcomes in Breast Cancer Survivors: A Randomized Controlled Trial
Article information
Abstract
OBJECTIVES
The number of breast cancer survivors throughout the world has increased. Breast cancer survivors need to know how to exercise to improve their clinical outcomes. The purpose of this study was to find the effects of an evidence-based combined exercise intervention on the fitness levels of breast cancer survivors.
METHODS
A total of 38 female breast cancer survivors were randomly assigned to an exercise group (n=19) or a control group who did usual care (n=19). Subjects in the exercise group participated eight weeks of moderate intensity aerobic exercise and bodyweight strength exercise three times per week. Weight, body mass index (BMI), cardiorespiratory fitness, handgrip strength, and flexibility were measured to assess the effects of the exercise intervention.
RESULTS
Women who completed the exercise intervention had decreased weight and BMI and increased cardiorespiratory fitness, handgrip strength, and flexibility.
CONCLUSIONS
Breast cancer survivors had improved health outcomes. A combined exercise intervention of moderate intensity three times per week for eight weeks can help breast cancer survivors improve their health. Participating in aerobic exercise and bodyweight strength exercise, which provide dynamic movements and use large muscle groups with no equipment, can help increase physical fitness levels of breast cancer survivors.
Introduction
Breast cancer is the most common women’s cancer and one of the main causes of cancer deaths in women, which was estimated at about 627,000 women or about 15% of all cancer deaths in women in 2018 [1]. Ninety percent of women patients with breast cancer had an average of 5-year survival rate [2] in 2018. Eighty percent of Korean women with breast cancer die within 5 years due to metastasis to other organs, regardless of breast cancer treatment. Recently, more women who had medical treatment for breast cancer have survived and it would be interesting to explore additional interventions, including exercise and physical activity, for improving breast cancer survivorship.
Participating in physical activity and exercise is strongly recommended for breast cancer patients and survivors. Several studies have found positive effects, including improving physical fitness, bone health, and quality of life, of physical activity and exercise on breast cancer survivorship [3, 4]. A recent meta-analysis found that moderate-intensity physical activity for 300 minutes per week can decrease both breast cancer-specific mortality and all-cause mortality [5]. Also, survivors of breast cancer who participated in diverse exercise interventions, including cardiorespiratory exercise and resistance exercise, improved their quality of life and physical fitness [6].
Considering the stages of the medical care and health condition of breast cancer patients, exercise intensity and amount have been varied in previous studies. The American College of Sports Medicine has provided exercise guidelines for breast cancer survivors [7] and has recommended 300 minutes per week of moderate intensity exercise. Previous studies reported that aerobic exercise or resistance exercise interventions help breast cancer survivors improve cardiovascular fitness, muscle strength, flexibility, quality of life, and physiological biomarkers of cancer, including breast cancer. While several studies reported the beneficial effects of combined aerobic and resistance exercise in cancer patients [8, 4, 9], there are a limited number of studies on exercise interventions in Korean cancer survivors. Also, bodyweight strength exercise consists of diverse movements using large muscle groups with minimal equipment and performed rhythmically that are easily performed by breast cancer survivors without specific skills [10, 11]. However, there is still a lack of evidence that the combined aerobic and strength exercise produces better outcomes in physical fitness in Korean breast cancer survivors.
We expected that a combined exercise intervention that includes aerobic exercise and bodyweight strength exercise might improve Korean breast cancer survivors’ cardiovascular fitness and muscular fitness. Therefore, the purpose of our research was to investigate the effects of combining aerobic and strength exercise to help improve breast cancer survivors’ cardiovascular fitness, muscle strength, and flexibility. Additionally, we conducted this evidence-based exercise intervention study to shed more light on the effects of this exercise approach in Korean breast cancer survivors.
Methods
Basic Characteristics of Participants
The participants were breast cancer survivors who were diagnosed with breast cancer in the past but were in remission. They had completed all their prescribed medical treatments, including chemotherapy, and had no recurrences of breast cancer for one year before the beginning of their participation in this study. All participants were recruited from breast cancer survivorship communities and local hospitals. They were randomly assigned to an exercise intervention group or a control group. The Institutional Review Board at XX University approved this study (7001988-201704-HR-171-02). All subjects agreed to participate in this study with a written informed consent form.
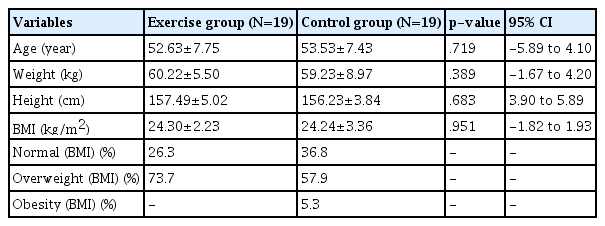
The inclusion criteria were completion of all medical treatments for breast cancer, no recurrence of breast cancer for at least one year, no diagnoses for other cancers or orthopedic conditions, approval from an oncologist in order to participate in exercise, and no contra-indications for physical activity. The exclusion criteria were having orthopedic or other pain and taking any kind of medications. Participants were excluded if they were taking any kind of medication. A total of thirty-eight breast cancer survivors were recruited from outpatient clinics at a local S hospital in Seoul, South Korea, throughout an eight-month period. The basic characteristics of the participants are in <Table 1>. We used a lottery system to randomize assignments into the exercise group or the control group. The exercise group participated in the exercise intervention and the control group had usual care. Participants of the control group received an exercise lesson at the end of the study if they completed the study and asked to get the exercise lesson. We measured only two times: before the intervention and after the intervention.
Measurements
We measured only two times: before the intervention and after the intervention. Trained exercise specialists assessed body weight, height, and physical fitness. We measured body weight and height using a calibrated Scale-Tronix scale (Scale-Tronix, White Plains, NY). We calculated BMI by dividing weight (kg) by the square of height (m2). The participants rested for 30 minutes after arriving to the research laboratory and before measuring resting heart rate.
We measured cardiorespiratory fitness with a three-minute step test. Participants stepped on a 12-inch box following a metronome beat set at 96 beats per minute for 3 minutes and sat on a chair for 1 minute immediately after to measure recovery heart rate [12]. The heart rate was measured during the first minute of recovery after the step test finished. We reported the instantaneous heart rate at the end of the 1-minute period post-exercise. Like other variables, we measured before and after the intervention.
Muscle strength was measured by the handgrip test, which positively correlates with upper-body strength [13]. Participants squeezed a dynamometer (T. K. K. 5401, Takei, Japan) for 3 seconds as much as they could exert themselves to determine maximal handgrip strength. The handgrip strength test was conducted twice on each hand, but the higher test score from each hand was recorded. We measured shoulder flexibility with the back scratch test that measures how close the hands can be touched together behind the back, one arm from above and one from below. The shoulder flexibility was performed with the right arm reaching from above and the left from below, and the left arm reaching from above and the right from below. We recorded the best performed score regardless of the measured arm.
Exercise interventions
The exercise group participated in an 8-week exercise program and the control group only had usual care for 8 weeks. <Table 1> describes the 8-week exercise intervention. The table shows the progression protocol, durations, intensities, modalities, and the selected exercise techniques. The exercise intervention consisted of combined aerobic and bodyweight strength exercise 3 times per week. The exercise group had a 5-minute dynamic warm-up followed by 15 minutes of moderate intensity treadmill running at 40-60% of the heart rate reserve (HRR), which is maximum heart rate minus resting heart rate. We calculated maximum heart rate as 220 minus age. We calculated exercise heart rate with the Karvonen formula: HRR * %intensity + HRrest, where HRrest is resting heart rate and %intensity is the prescribed exercise intensity (40-60%). The exercise intensity followed the guidelines in <Table 3>. Heart rate was palpated on the left arm by a researcher for a period of 15 seconds in the middle of the exercise sessions. The bodyweight strength exercise consisted of 3 sets of 8 repetitions in the first week, 3 sets of 10 repetitions in the second and third weeks, and 3 sets of 12 repetitions from the fourth to the eighth week. The bodyweight exercises were lunges, burpees, jumping jacks, mountain climbers, Superman, crunches, and squats, in that order. We used a whole body routine for the bodyweight strength exercise sessions and no equipment. Participants had a 30-second break between sets.
Statistical analysis
We used the SPSS 25.0 software (SPSS Inc., Chicago, IL, USA) to analyze our data. All values are presented as mean ± standard deviation (SD). Descriptive statistics include means and SD. Independent t-test was used to compare the Basic Characteristics of Participants. Continuous data were tested for normality using the Shapiro-Wilk test. A 2X2 (group x time) mixed ANOVA model with a repeated factor was used to evaluate group differences and compare pre and post 8-week exercise intervention data with control. A simple effect test was conducted to examine specific difference in pre-post scores of individual group when significant interaction effect was found. A p-value less than 0.05 was considered statistically significant. We also reported effect size (Eta Squared). The Eta squared is the proportion of variance associated with one or more main effects, errors or interactions in ANOVA. An effect size of less than 0.2 was considered small, 0.5 was considered medium, and 0.8 was considered large.
Results
Baseline characteristics of study participants
<Table 2> presents basic anthropometric characteristics of the participants. Frothy breast cancer survivors were recruited for this study, but two participants dropped out at their own request. A total of thirty-eight participants completed the study. Nineteen participants were in the exercise intervention group and nineteen were in the control group. We did not find any significant differences on baseline measures between the control and the intervention groups.
Inferential statistical tests
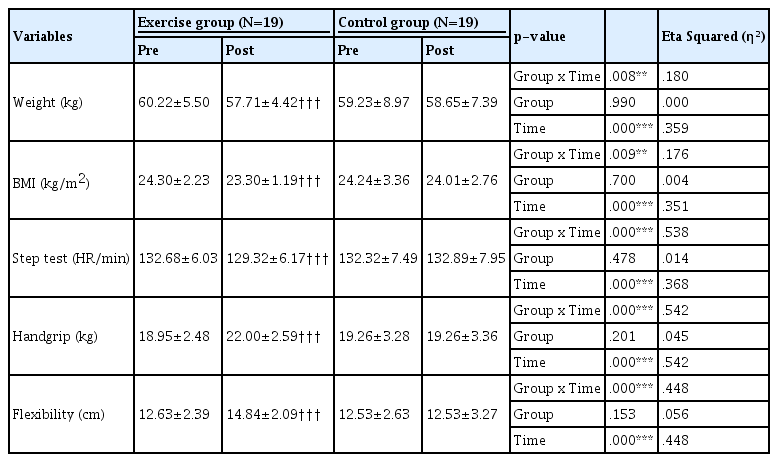
Results of 2X2 ANOVA were shown by <Table 3>. There were significant interaction effects of body weight, BMI, cardiorespiratory fitness, handgrip strength, and flexibility.
Body weight and BMI
For intervention group, post-test in body weight (57.71±4.42 kg, p<0.001) and BMI (23.30±1.19 kg/m2, p<0.001) were significantly lower than pre-test in body weight (60.22±5.50 kg) and BMI (24.30±2.23 kg/m2). For control group in body weight and BMI there were no difference between pre-test and post-test.
Measurements of cardiorespiratory fitness, handgrip strength, and flexibility
For intervention group, post-test in heart rate (129.32±6.17 bpm, p<0.001) was significantly lower than pre-test in heart rate (132.68±6.03 bpm). Also, for intervention group, post-test in handgrip strength (22.00±2.59 kg, p<0.001), and flexibility (14.84±2.09 kg, p<0.001) were significantly higher than pretest in handgrip strength (18.95±2.48 kg), and flexibility (12.63±2.39 kg). For control group in heart rate at the end of the step test, handgrip strength, and flexibility, there were no difference between pre-test and post-test. Effect sizes in heart rate, handgrip and flexibility were medium.
Discussion
Breast cancer survivors who participated in an 8-week evidence-based combined exercise intervention achieved positive outcomes regarding cardiorespiratory fitness, handgrip strength, and shoulder flexibility compared to a usual care control group. Most breast cancer survivors, who complete their medical treatments, wonder about what is the best way to prevent the breast cancer from recurring even though their oncologists already recommended them several beneficial health behaviors, including participating in physical activity and exercise. Our study investigated the beneficial effects of an evidence-based exercise intervention combining aerobic exercise and bodyweight strength exercise that can improve cardiorespiratory fitness and whole body strength. The reason for the lack of results in the control group was related to the fact that the participants in the control group underwent their usual care and kept their daily activity the same as since completed their oncological treatments. While obesity can have a protective effect in cancer patients, our exercise group participants were normal weight and only 5.3% of the participants in the control group were obese, throughout the study. Weight loss is preferred under many circumstances, but it is most important to maintain a normal weight in cancer survivors [14].
Participating in the exercise intervention showed beneficial effects in cardiorespiratory fitness and handgrip strength. Our findings coincided with previous exercise intervention studies [15, 16] that used aerobic exercise and resistance exercise. Women who completed the exercise intervention in our study had decreased recovery heart rate post-intervention that may be related to the lower body weight. Previous studies have reported that higher cardiorespiratory fitness levels may be associated with decreased recurrence of breast cancer and mortality [17, 18]. Also, women in the exercise group had increased handgrip strength post-intervention, which is an indirect indicator of upper-body muscle strength. Women in our exercise group improved both cardiorespiratory fitness and muscular fitness, which help improve overall fitness levels.
The exercise intervention helped increase shoulder flexibility, even though flexibility exercises were not performed. We performed the back scratch test on the right and left shoulders. We then used the higher score regardless of whether the more flexible side was the side of treatment or both sides were treated. The participants generally have uncomfortable shoulder and arm movements after medical treatments for breast cancer such as surgery. Increasing shoulder and arm flexibility is important for improving daily movement. Increased shoulder flexibility measured by the back scratch test is associated with increased range of motion, which helps shoulder and arm movements [19]. A previous meta-analysis reported that an exercise intervention including aerobic exercise, resistance exercise, yoga, and Pilates increased shoulder flexibility, mobility, and range of motion of the upper-body [20, 21].
In this study, intervention strategy was to provide moderate intensity exercise that combined aerobic and bodyweight strength exercise to women who were breast cancer survivors. Recent studies also recommend breast cancer survivors to participate in aerobic and resistance exercise with intensities higher than moderate [22]. Our exercise intervention has positive results in body weight reduction and physical fitness improvement. The exercise frequency in our study was three times per week with one recovery day between sessions, replicating previous studies [23, 24]. Bodyweight strength exercise may be a better alternative to traditional weight lifting resistance exercise for breast cancer survivors in the long term. It can be implemented as a home-based exercise approach because of the lower chance of injury than traditional weight lifting resistance exercise [20] and with the need for limited or no equipment. Previous studies also demonstrated that bodyweight circuit strength exercise reduced weight and accumulated fat and increased physical fitness in obese and cancer patients [25].
We can suggest possible mechanisms for the beneficial effects of our exercise intervention. Regular exercise helps reduce insulin resistance [26] and alters adipocytokines by raising adiponectin and decreasing leptin and inflammation [27, 28]. Those physiological changes are associated with reduced body weight to a similar extent as what we achieved with our exercise intervention. Lastly, our intervention protocol involves the combination of exercises with movement patterns of spontaneous ranges of motion that allows the participants to benefit indirectly with increased flexibility.
There are several limitations in this study. First, the sample size of this study is too small to generalize our findings over entire populations. Second, we did not make sure the participants had consistent daily physical activity and diets, although we encouraged them to have the same meals and amount and type of non-exercise physical activity as usual during the study. Third, the duration of our exercise intervention was relatively short; longer trials are needed to further clarify the effects of different exercise interventions.
Conclusions
Our exercise intervention was a moderate intensity, combined aerobic and bodyweight strength exercise. After 8-week exercise intervention, there are positive outcomes in decreasing body weight and BMI and increasing cardiorespiratory fitness, handgrip strength, and upper-body flexibility. Further studies are needed to understand the effects of exercise interventions on the physiological and biological changes that improve breast cancer survivors’ health condition and to provide guidelines for alternate and targeted exercise recommendations.
Acknowledgements
This work was supported in part by the National Research Foundation of Korea (NRF-2015S1A5B5A01015881).
Notes
Declare conflicts of interest or state “The authors declare no conflict of interest.” Authors must identify and declare any personal circumstances or interest that may be perceived as inappropriately influencing the representation or interpretation of reported research results. Any role of the funding sponsors in the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript, or in the decision to publish the results must be declared in this section. If there is no role, please state “The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results”.